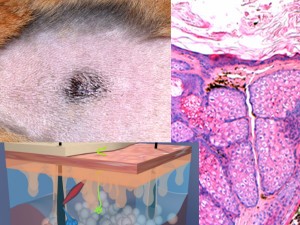
Hamster Flank Organ Acne Model
Introduction
Hamster flank organ acne model is an animal model which has been extensively used for the study of compounds inhibiting the response of sebaceous glands to hormones, anti-androgens, and retinoids.
The hamster flank organ is composed of clusters of large sebaceous glands.
This organ has a bilateral localization on animals abdomen and is used to produce pheromones and marking molecules of mature males.
Protocol summary
– Hamster mature males with application of compounds / new drugs on flank organ on each side.
– Histology evaluation flank organ size and sebaceous glands.
– Evaluation of in vivo / ex vivo / analysis secondary efficacy endpoints
Typical Results
| Evaluation | Parameter | Group control (excipient / topical) | Test group (retinoid / topical) |
|---|---|---|---|
| In vivo | Bodyweight | NE (No Effect) | NE or decrease |
| Flank organ size | NE | Slight decrease | |
| Back skin scaling (visual scoring) | NE | Slight increase (dry skin appearance) | |
| Back skin erythema (visual scoring) | NE | Increase (irritation) | |
| Back skin thickness (calipering) | NE | NE or increase | |
| Epidermis thickness (OCT / Optical Coherence Tomography) | NE | Increase | |
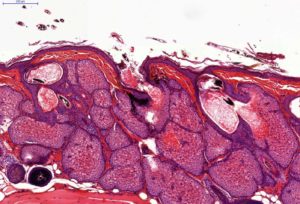
| Ex vivo (histology) | Sebaceous glands | NE | Decrease |
| Sebum surface | NE | Decrease | |
| Sebaceous glands inter-lobular spaces | NE | Increase | |
| Epidermis thickness | NE | Increase (epidermal hyperplasia) |
|
| Dermis inflammation | NE | Increase (local inflammation slight to moderate) |
Hamster Flank Organ Acne Model / Detailed protocol
Hamster / Syrian gold strain:
– >10 weeks old / Male with SPF status
– Randomization on bodyweight & organs sizes
Application of dermatological pharmaceutical products:
– Ready to use formulations / Formulation screening on request
– Topical & Oral routes / Reference drugs: Isotretinoin or Adapalene or Bexarotene
– Intra-dermal route (i.e. Medical Devices or injectable drugs)
– Treatment duration: >3 weeks
Evaluation of clinical macroscopic signs:
– Body weight / General behavior
– Flank organ size and length / Caliper
– Back skin thickness / Caliper
– Flank organ and surrounding skin macroscopic description:
Scaling (skin dryness)
Erythema (skin redness)
Other macroscopic skin observations

In vivo imaging:
– Macroscopical observation: Digital pictures of flank organs
– Sub-macroscopical exploration: Optical Coherence Tomography (OCT) for epidermis thickness measurement and superior sebaceous gland structure (on development)
– Ultrasonography for flank organ size evaluation
Ex vivo imaging:
– Histology on back skin slices obtained from paraffin blocks or frozen blocks
– HES (Hemalun-Eosin-Safran) / Overall evaluation of efficacy (sebaceous glands surface / sebum surface)
– Immuno-histochemistry / IHC on request
Other ex vivo imaging techniques of interest:
– 3D reconstruction of sebaceous glands for volume evaluation
– Light Sheet microscopy (in collaboration)
– MALDI tissue imaging (in collaboration)
Hamster Flank Organ Acne model / Conclusion
Hamster flank organ model is a first intention model validated anti-androgens family of compounds.
6 to 8 animals per group are generally sufficient to underline anti-acne effect of new therapeutics (based on difference of at least 20% / sebaceous glands surface or sebum surface).
Do not hesitate to contact us if you need more information or a quotation on this model.
Bibliography of interest
- Franz, T. J., et al. The hamster flank organ model: is it relevant to man? J. Invest. Dermatol. 93, 475–9 (1989).
- Weissmann, A., et al. Morphometric Studies of the Hamster Flank Organ: An Improved Model to Evaluate Pharmacologic Effects on Sebaceous Glands. J. Invest. Dermatol. 82, 522–525 (1984).
- Gomez, E. C. & Moskowitz, R. J. Effect of 13-cis-retinoic acid on the hamster flank organ. J. Invest. Dermatol. 74, 392–397 (1980).
- Orentreich, N. et al. The local antiandrogenic effect of the intracutaneous injection of progesterone in the flank organ of sexually mature male Syrian golden hamster. Arch. Dermatol. Res. 276, 401–405 (1984).
- Piskin, S. & Uzunali, E. A review of the use of adapalene for the treatment of acne vulgaris. Ther. Clin. Risk Manag. 3, 621–624 (2007).